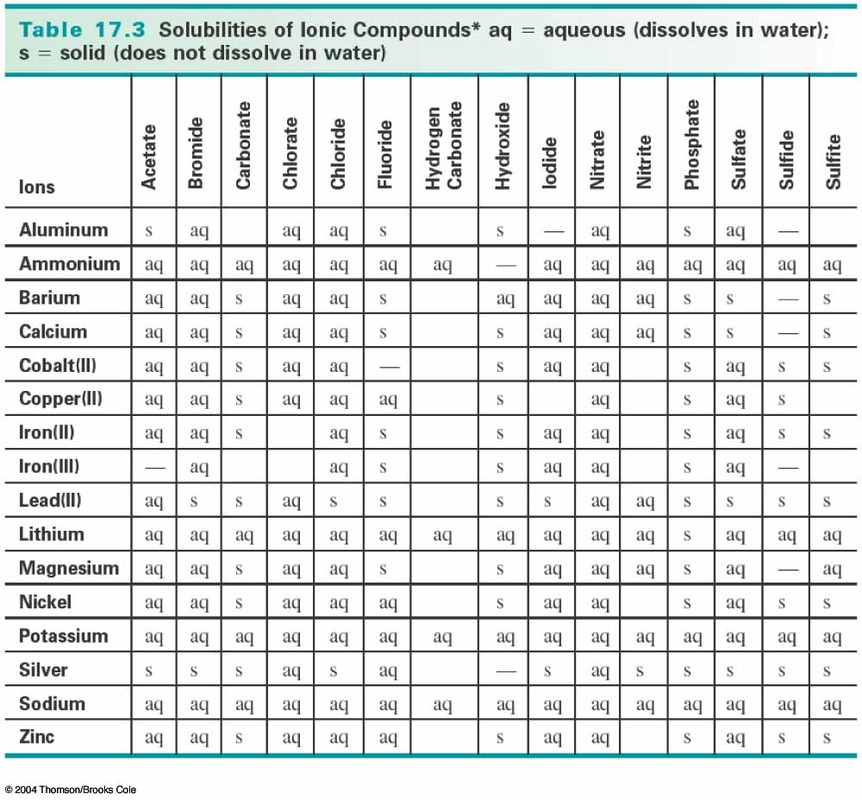
We cannot see the ammonium cations or the chloride anions dissolved. Once again, the ammonium chloride solution just looks like water. Next I'm pipetting out some of the ammonium chloride solution. Once again you can see that that is a clear, colorless and homogeneous solution. First, I'm pipetting out some of the sodium phosphate solution. Now let's test your prediction by doing an actual experiment. Feel free to look at the solubility rules as you try to solve this problem. 
So the real question we need to ask ourselves is, does sodium chloride or ammonium phosphate precipitate? Go ahead and try to answer that on your own now. 
In the containers, I have four different ions, sodium, phosphate, ammonium, and chloride. Let's mix these two solutions and predict what will happen. Since the ammonium chloride was very highly soluble in the water, the solubility product constant must be a very large value. In this case, the exponents are both one, because in the balanced chemical reaction equation, all of the coefficients are one. It's the molar concentration of ammonium, ion, times the molar concentration of chloride ion. Leaving us with this expression for the solubility product constant. The solubility product constant must be the product of the cation times the anion divided by the reactant, which in this case is once again a solid. Once again, we can write the equilibrium constant for this reaction. Can you remember what the chemical formula is for ammonium chloride? Well done, when the ammonium chloride dissolves, in the water, that makes ammonium cation and chloride anion. And in container B this time, we're going to have an ammonium chloride solution. So first we're going to have, in container A, our sodium phosphate solution. But we'll be mixing the sodium phosphate solution with other types of solutions. We're going to continue doing that in this lecture. In the last lecture we mixed a solution of sodium phosphate with another solution. We will learn how to apply our solubility rules and identify the spectator ions. And writing the balanced net ionic equation that results. Determining if a product will precipitate when two aqueous solutions are mixed. Writing balanced solution chemical reaction equations. In this lecture, we will continue practicing several skills.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
